Treating Cerebral ALD
Treating Cerebral ALD
Allogeneic hematopoietic stem cell transplantation (allo-HSCT)
Currently, the only treatment for cerebral adrenoleukodystrophy (ALD) that can stabilize disease progression is allogeneic hematopoietic stem cell transplantation (allo-HSCT)—and it can be lifesaving.1 Early diagnosis during the first signs of cerebral involvement is critical because outcomes with allo-HSCT are more favorable with earlier initiation of treatment.2
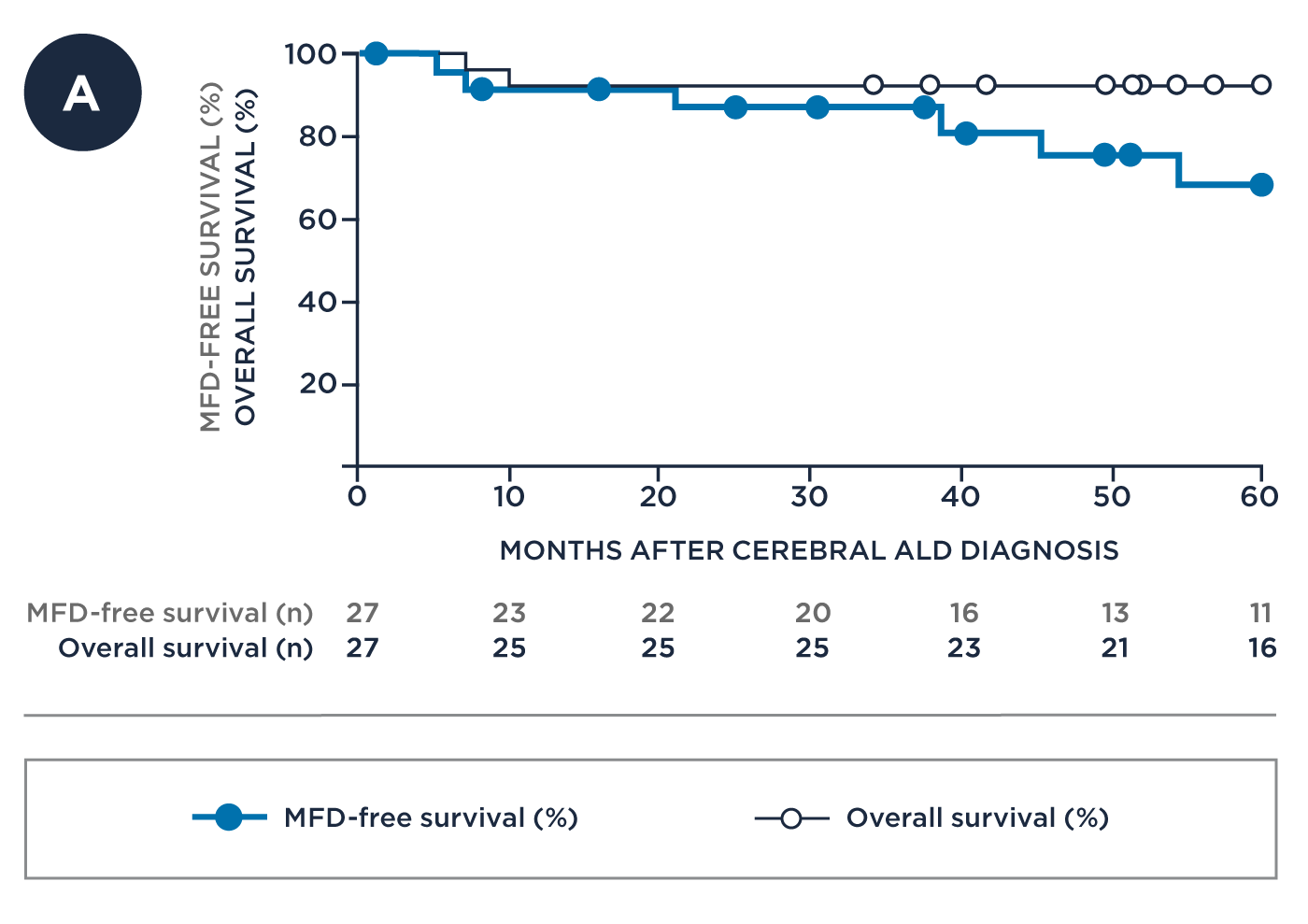
In a study of patients with both early and advanced cerebral ALD*
Treatment early in the course of cerebral disease demonstrated greater major function disability (MFD)†-free survival and overall survival3
| MFD-FREE SURVIVAL | OVERALL SURVIVAL‡ |
||
|---|---|---|---|
| 2 YEARS2 YRS | 5 YEARS5 YRS | ||
| Early Disease* (n=27) | 91% | 76% | 94% |
| Advanced Disease* (n=10) | 20% | 10% | 90% |
| MFD-FREE SURVIVAL |
OVERALL SURVIVAL‡ |
||
|---|---|---|---|
| 2 YRS |
5 YRS |
||
| Early Disease* (n=27) |
91% | 76% | 94% |
| Advanced Disease* (n=10) |
20% | 10% | 90% |
‡Overall survival was assessed at both 2 and 5 years from cerebral ALD diagnosis.
Early Disease* with Number of Patients at Risk
Advanced Disease* with Number of Patients at Risk
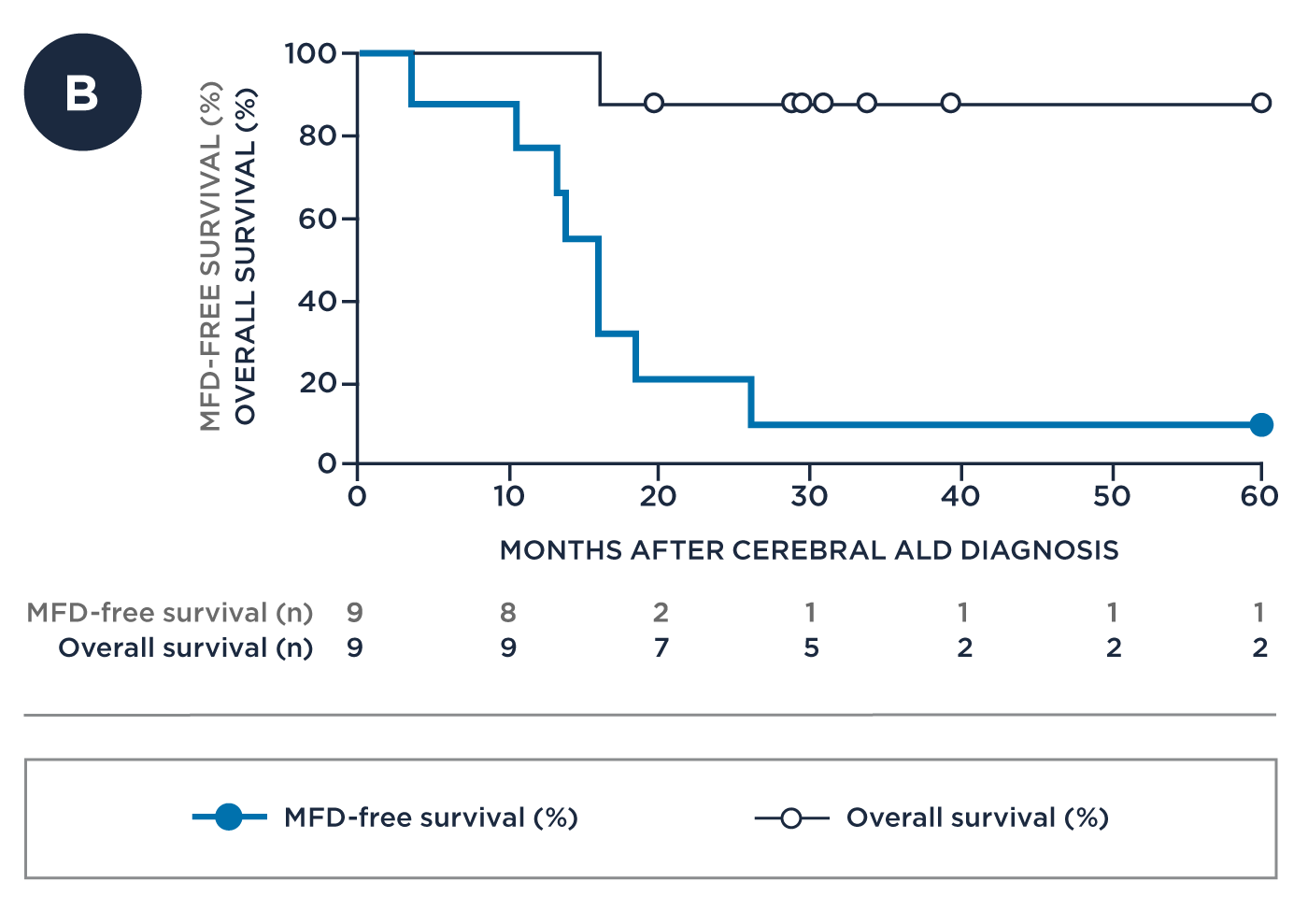
* Early disease: NFS≤1, Loes score 0.5 to ≤9, GdE+; Advanced disease: NFS>1, Loes score >9, GdE+; NFS=neurologic function score: a 25-point score used to evaluate the severity of gross neurologic dysfunction in cerebral ALD by scoring 15 symptoms across 6 categories (hearing, communication, vision, feeding, locomotion, and incontinence); Loes score is a 34-point scale commonly used to quantify the extent of demyelinating brain lesions seen on MRIs in cerebral ALD (higher scores indicate more severe disease); GdE+=positive for gadolinium contrast enhancement, an indicator of active neuroinflammatory disease.
† MFDs: loss of communication, cortical blindness, tube feeding, total incontinence, wheelchair dependence, and complete loss of voluntary movement.
In addition to measuring overall survival, it is also important to measure MFD-free survival in cerebral ALD. MFDs are six severe disabilities commonly attributed to cerebral ALD-they are of particular clinical importance because they can significantly compromise a patient’s ability to function independently. These MFDs are:3
- Loss of communication
- Cortical blindness
- Tube feeding
- Total incontinence
- Wheelchair dependence
- Complete loss of voluntary movement
Although allo-HSCT has survival benefits, it also has significant risks and challenges1,3,4
Allo-HSCT can stabilize disease progression if performed during the early stage of cerebral involvement, but it has significant associated risks. These include transplant-related mortality (TRM), graft failure or rejection, graft-versus-host disease (GVHD), and the potential for opportunistic infections.1,3,4
- 100-day transplant-related mortality (TRM) rates of 8% to 12% have been reported for patients receiving allo-HSCT1,3,4
- Engraftment after allogeneic transplant is not a certainty, with graft failure rates ranging from 5% to 18%1,3,4
- The development of acute and chronic graft-versus-host disease (GVHD) is a significant risk of allogeneic transplant leading to long-term morbidity and significant decrease in survival. GVHD rates have been reported at 18% to 39% for acute GVHD grades 2-4 and 7% to 32% for chronic GVHD1,3,4
- The need for immunosuppression to prevent or treat GVHD following allo-HSCT is also associated with a prolonged, continuous risk, including serious infection which has been reported in 11% to 29% of patients3-7
Safety outcomes are typically more favorable if allo-HSCT is performed using cells from a human leukocyte antigen (HLA)–matched sibling donor; however, less than 30% of patients have a matched sibling donor.1,3
Early detection can save lives
We want to be able to identify progression early so that we can recognize the condition in time for the patient to benefit from the treatment.
Bradley Miller, MD, PhD Paediatric endocrinologist
Endocrinologists are often the first line of care for ALD patients, and have an opportunity to diagnose ALD early.2,8
Test to detect
Measuring very-long chain fatty acids (VLCFAs) levels can help save lives2,9
1. Miller WP, Rothman SM, Nascene D, et al. Outcomes after allogeneic hematopoietic cell transplantation for childhood cerebral adrenoleukodystrophy: the largest single-institution cohort report. Blood. 2011;118(7):1971-1978. 2. Moser HW, Mahmood A, Raymond GV. X-linked adrenoleukodystrophy. Nat Clin Pract Neurol. 2007;3(3):140-151. 3. Raymond GV, Aubourg P, Paker A, et al. Survival and Functional Outcomes in Boys with Cerebral Adrenoleukodystrophy with and without Hematopoietic Stem Cell Transplantation. Biol Blood Marrow Transplant. 2019;25(3):538-548. 4. Mitchell R, Nivison-Smith I, Anazodo A, et al. Outcomes of haematopoietic stem cell transplantation for inherited metabolic disorders: a report from the Australian and New Zealand Children’s Haematology Oncology Group and the Australasian Bone Marrow Transplant Recipient Registry. Pediatr Transplant. 2013;17(6):582-588. 5. Reddy SM, Winston DJ, Territo MC, Schiller GJ. CMV central nervous system disease in stem-cell transplant recipients: an increasing complication of drug-resistant CMV infection and protracted immunodeficiency. Bone Marrow Transplant. 2010;45:979-84. 6. Inagaki J, Noguchi M, Kurauchi K, et al. Effect of cytomegalovirus reactivation on relapse after allogeneic hematopoietic stem cell transplantation in pediatric acute leukemia. Biol Blood Marrow Transplant. 2016;22:300-6. 7. Garcia-Cadenas I, Rivera I, Martino R, et al. Patterns of infection and infection-related mortality in patients with steroid-refractory acute graft versus host disease. Bone Marrow Transplant. 2017;52:107-13. 8. Bornstein SR, Allolio B, Arlt W, et al. Diagnosis and Treatment of Primary Adrenal Insufficiency: An Endocrine Society Clinical Practice Guideline. J Clin Endocrinol Metab. 2016;101(2):364-389. 9. Engelen M, Kemp S, Poll-The BT. X-linked adrenoleukodystrophy: pathogenesis and treatment. Curr Neurol Neurosci Rep. 2014;14(10):486.
